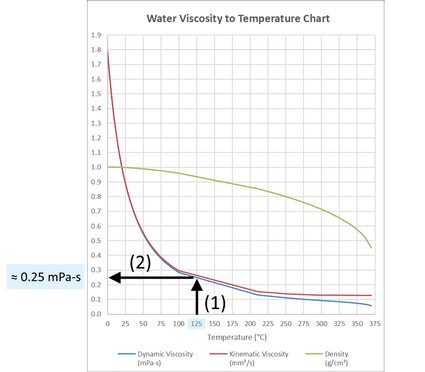
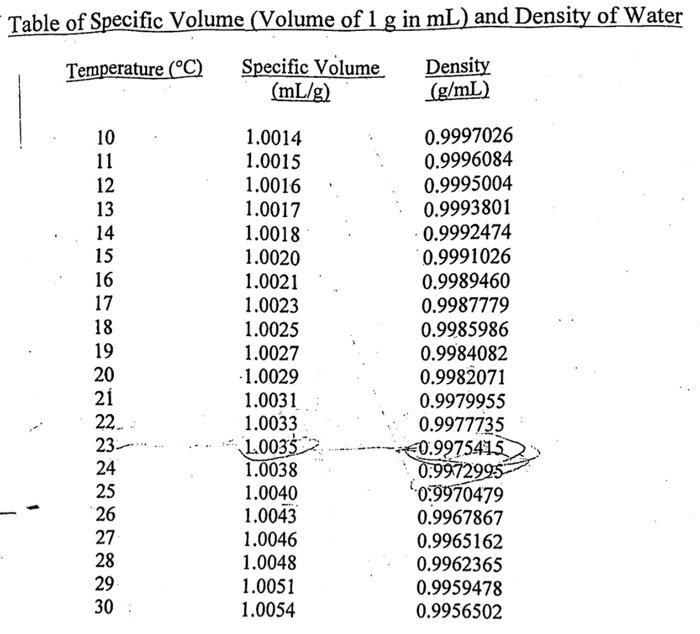
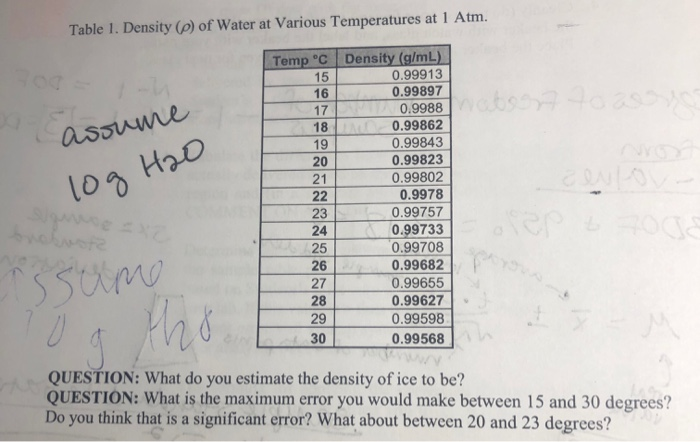
The dissociation of water at 25^∘ C is 1.9 × 10^-7% and the density of water is 1.0 g/cc. The ionisation constant of water is :

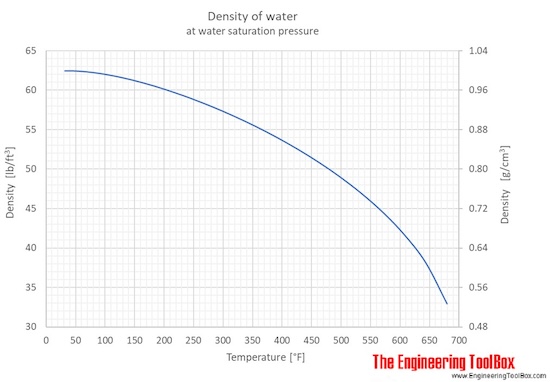
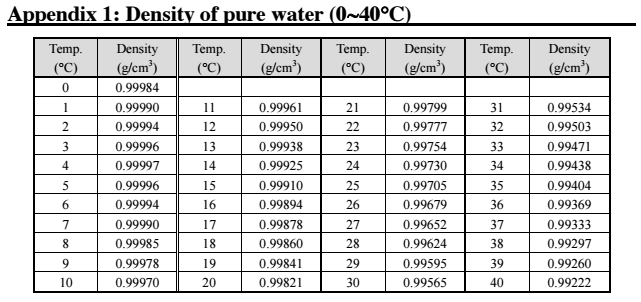
SOLVED:At 25^∘ C the density of water is 0.997 g / cm^3, whereas the density of ice at -10^∘ C is 0.917 g / cm^3 . (a) If a soft-drink can (volume =
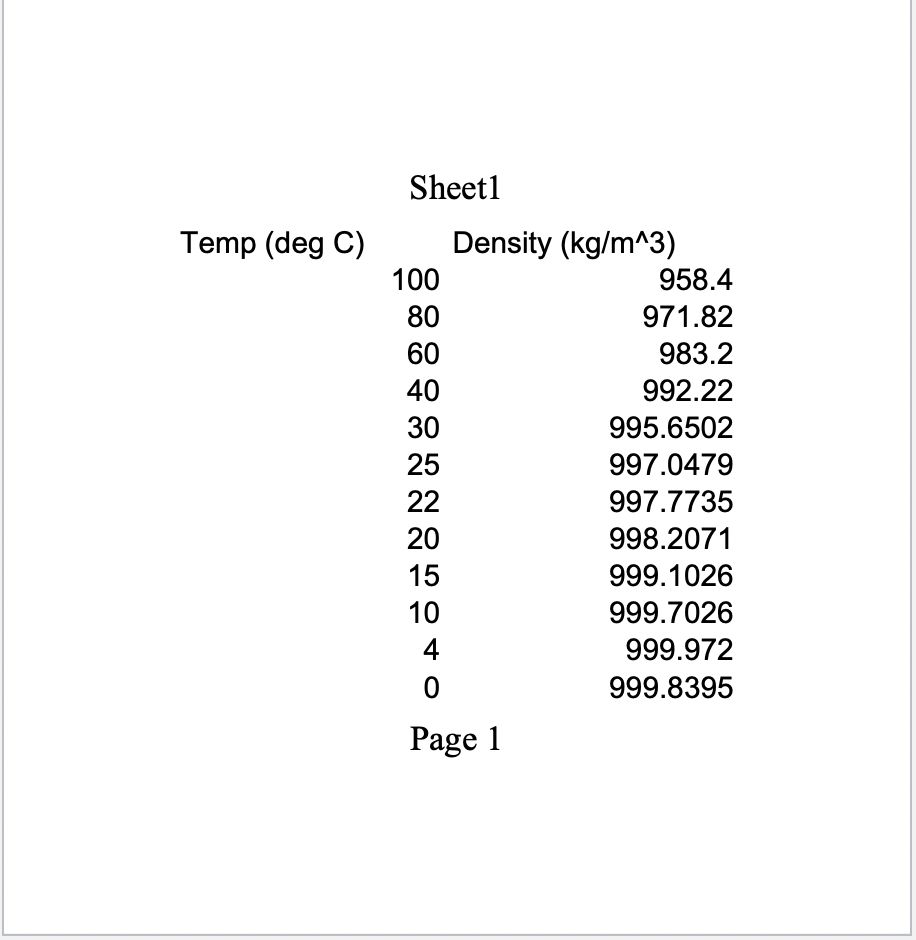
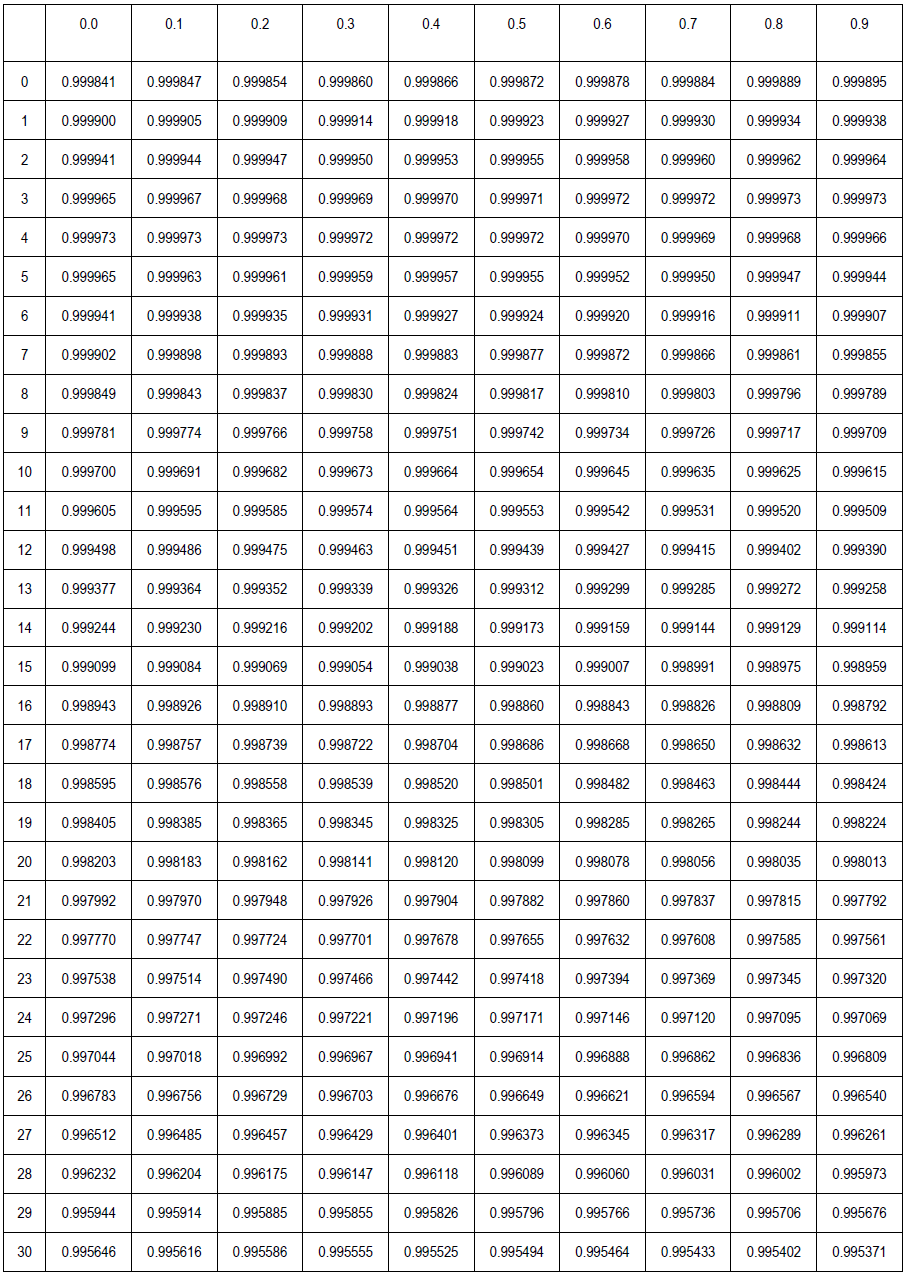
Density of Water (g/cm3) at Temperatures from 0°C (liquid state) to 30.9°C by 0.1°C increments. 0.0 0.1 0.2 0.3 0.4
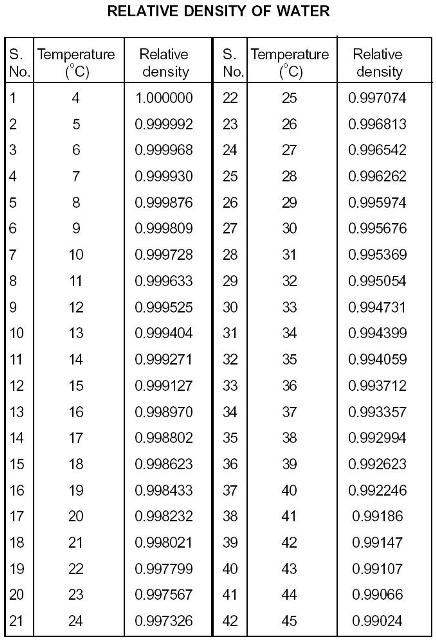
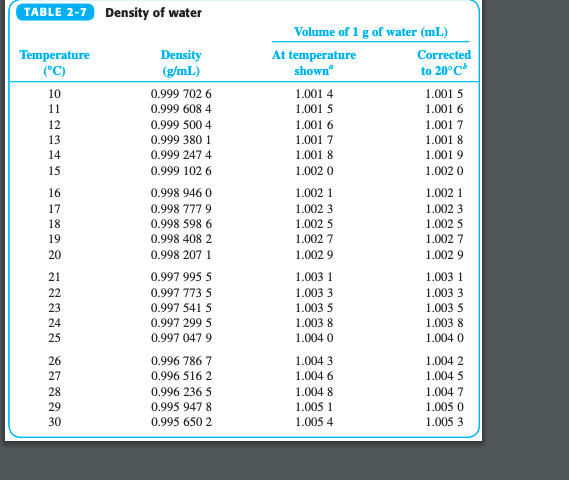
Relative Density Of Water - Civil Engineering Portal - Biggest Civil Engineering Information Sharing Website