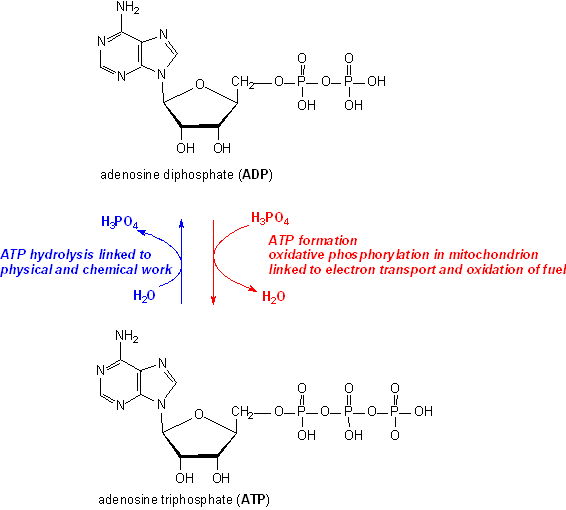
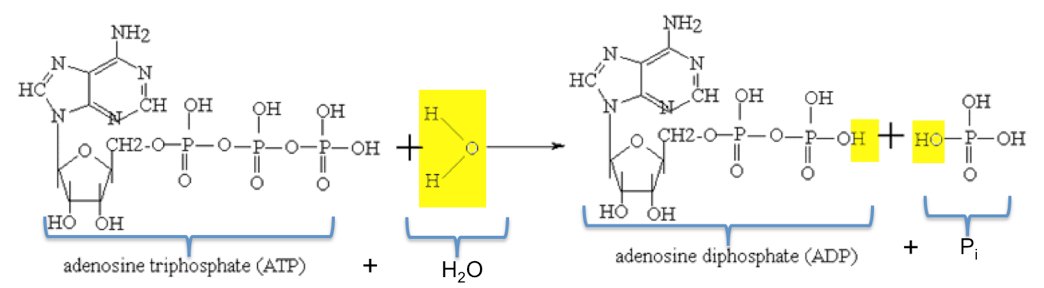
A water molecule is added to an atp molecule to break atp down into adp and a phosphate group. write the - Brainly.com
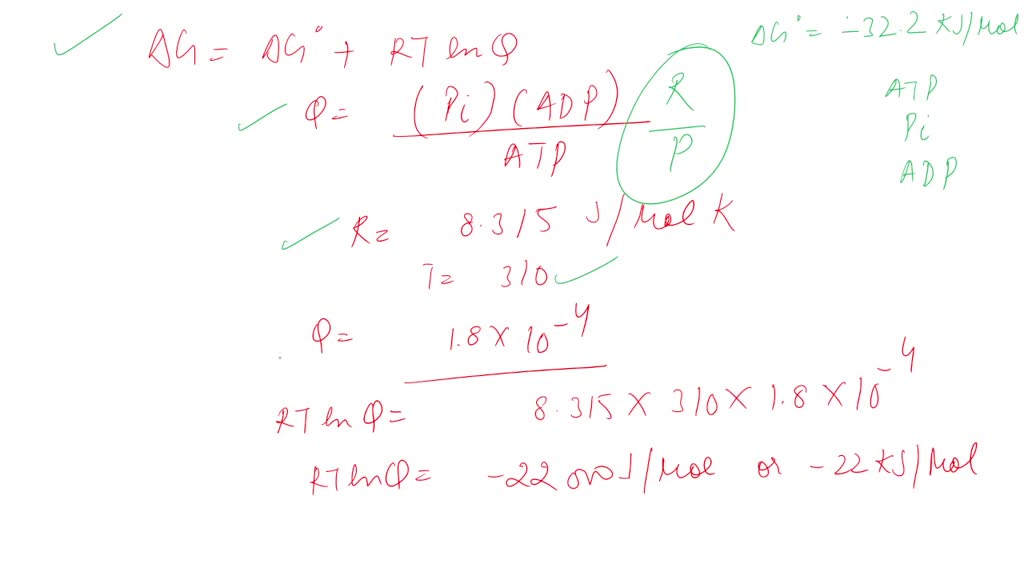
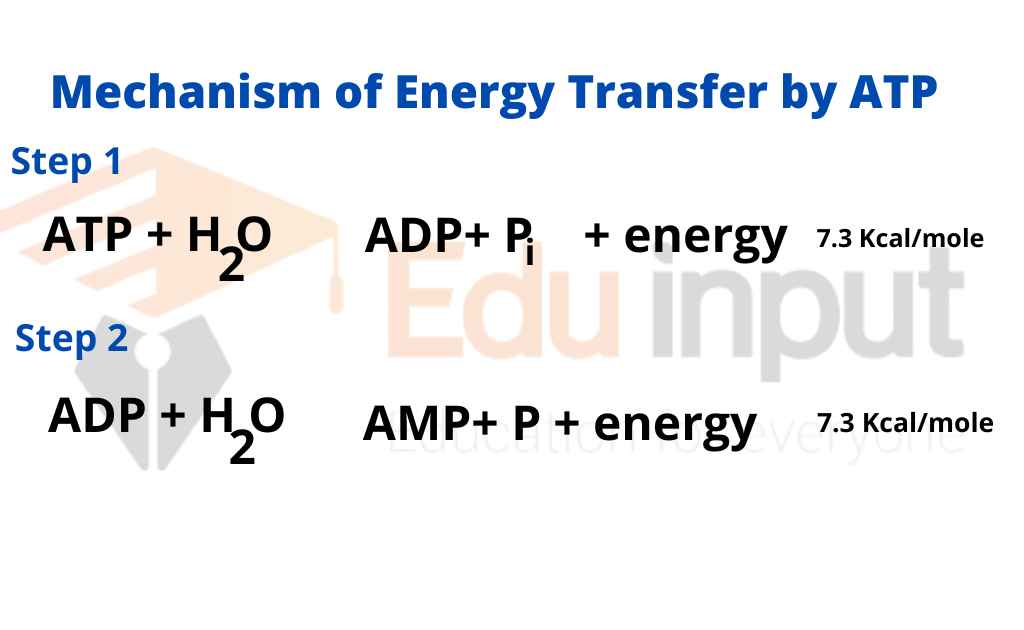
SOLVED: The standard free energy of hydrolysis (∆G°´) of ATP is -32.2 kJ/mol. ATP + H2O → ADP + Pi The actual free energy of ATP hydrolysis, represented by ∆G, depends on
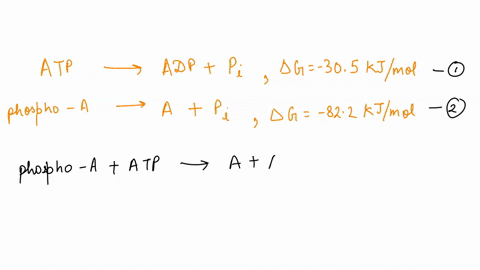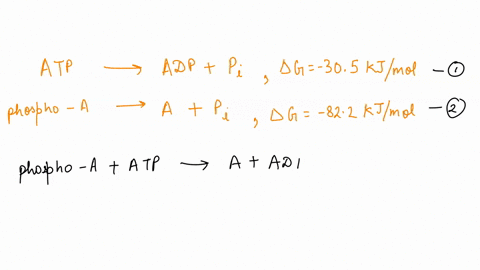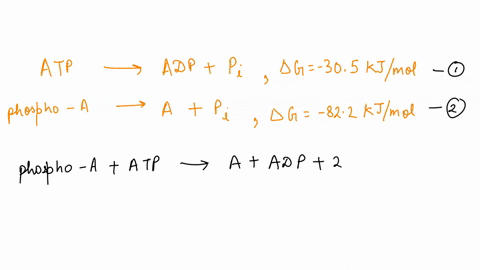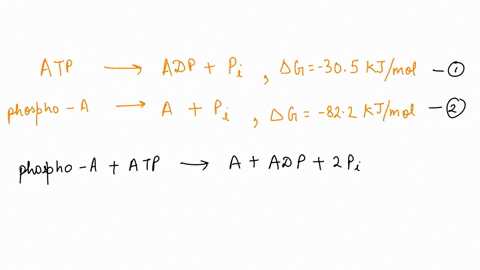
SOLVED: ATP + H2O <—> ADP + Pi Calculate the ΔG for ATP hydrolysis when the concentrations of ATP, ADP, and Pi are all 1M R = 1.98 x 10-3 T = 310 kelvin ΔGº = -30.5 kJ/mol




.PNG)


![Solved ATP + H2O → ADP + Pi [AGO' = -7.3 kcal/mol ] Question | Chegg.com Solved ATP + H2O → ADP + Pi [AGO' = -7.3 kcal/mol ] Question | Chegg.com](https://media.cheggcdn.com/media/c65/c65cc5ef-e21f-4e67-8f65-9edf4841d0ef/phpTtmcCV)
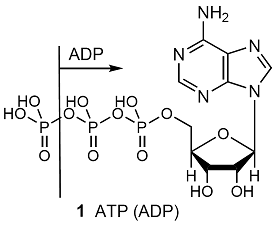

![Energy in breaking bonds [ATP and ADP]? | ResearchGate Energy in breaking bonds [ATP and ADP]? | ResearchGate](https://www.researchgate.net/profile/Yufei_Huang20/post/Energy_in_breaking_bonds_ATP_and_ADP/attachment/5e525ed5cfe4a7bbe5617e1e/AS%3A861699309584385%401582456533095/download/IMG_4426.jpg)