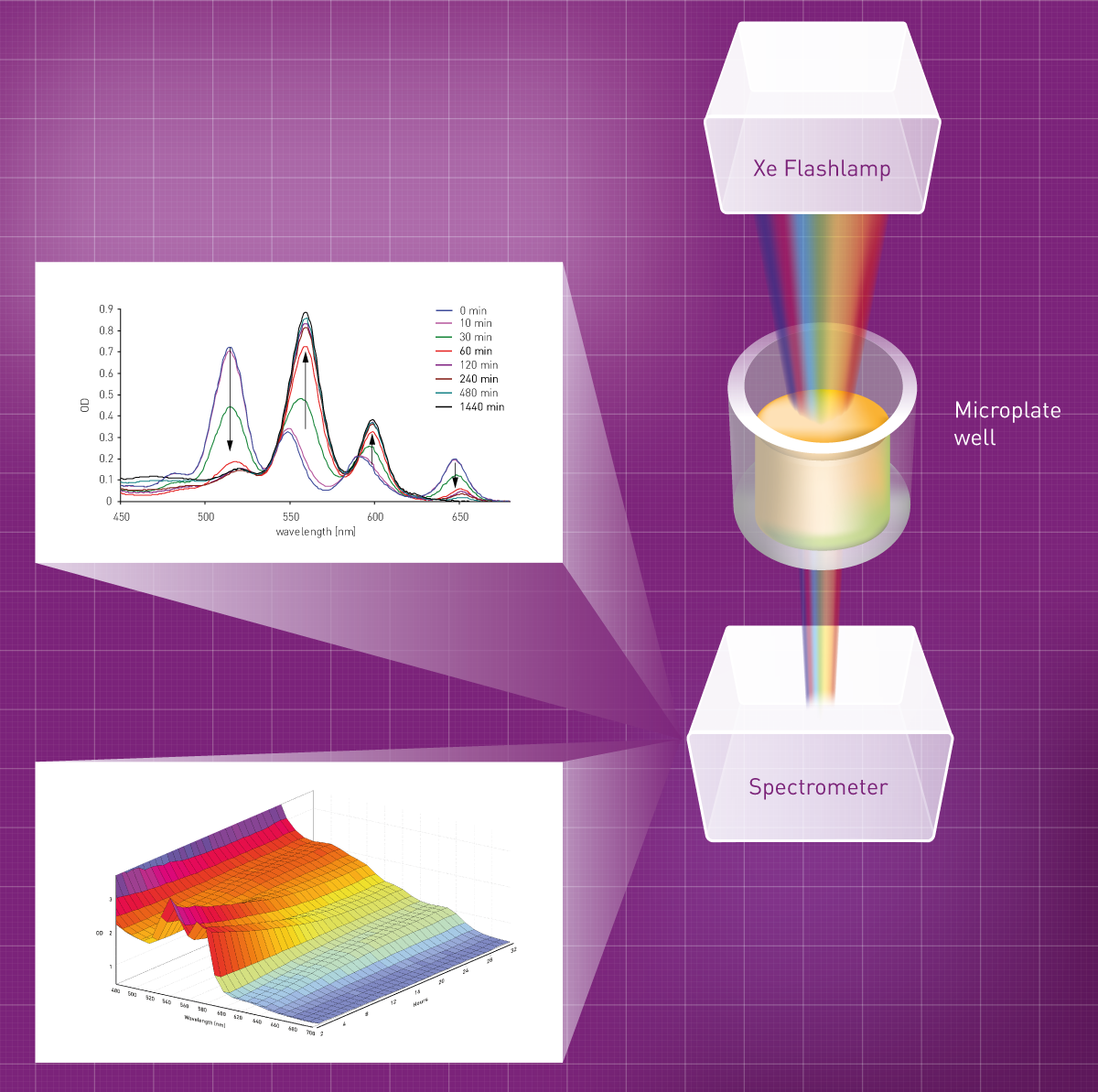
Absorbance Measurements – the Quick Way to Determine Sample Concentration - Eppendorf Handling Solutions

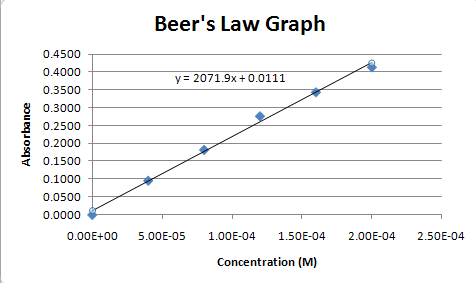
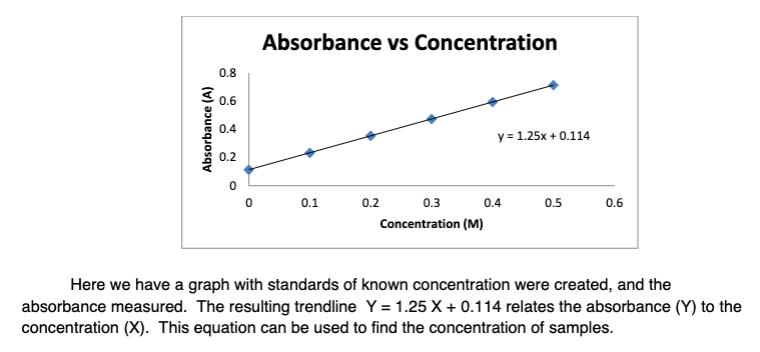
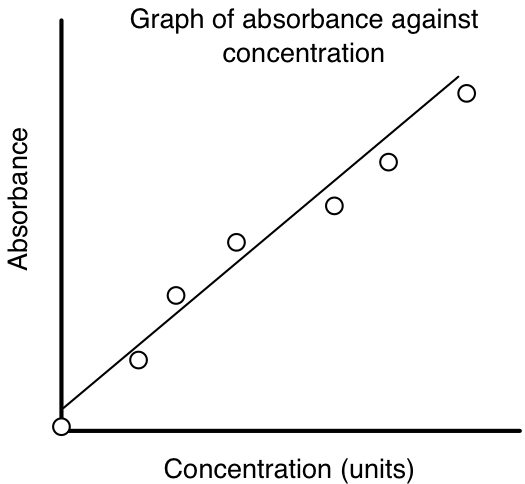
Use the data to plot a graph of Absorbance versus Concentration. Determine the concentration of the dye which would correspond to an absorbance of 0.140. What does Beer's Law say about the
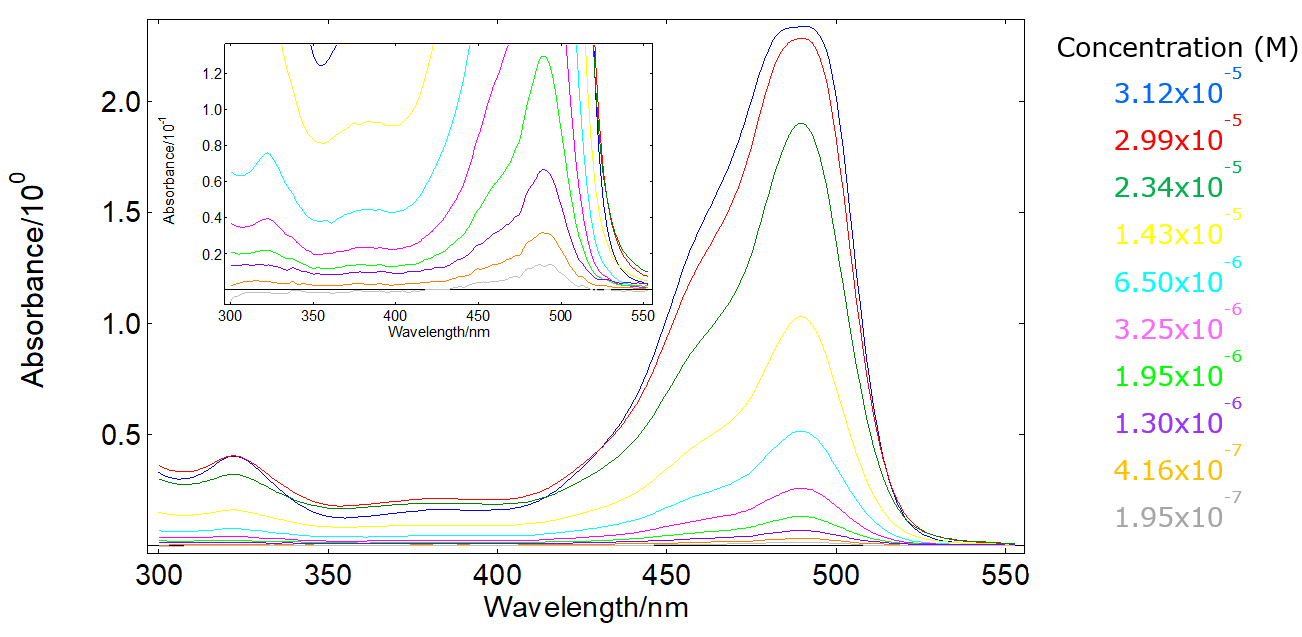
Absorbance (A) versus wavelength (λ) of Ni0.92Co0.08O thin films at... | Download Scientific Diagram

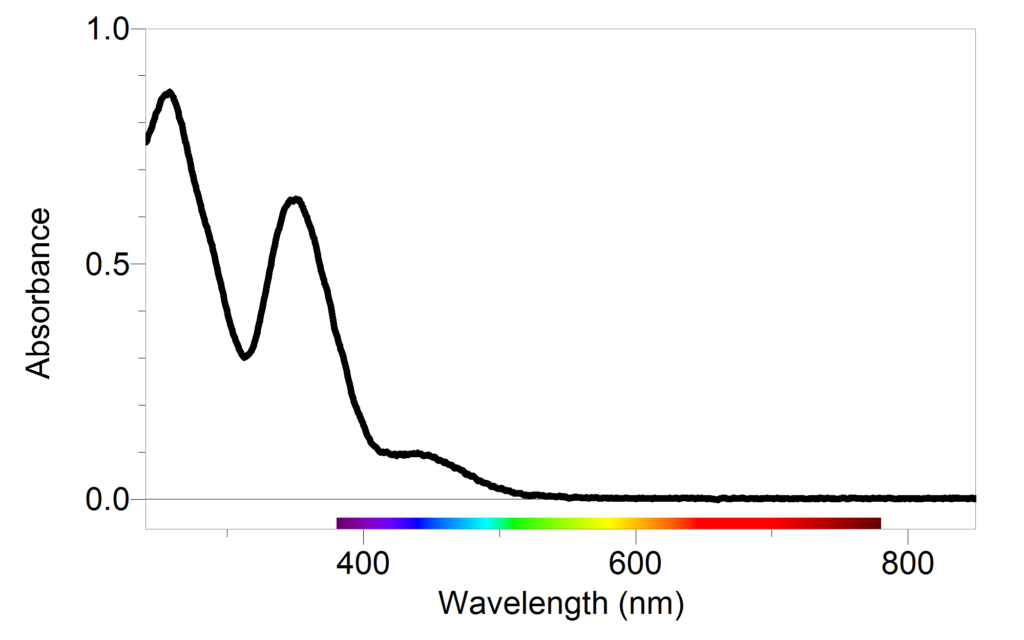
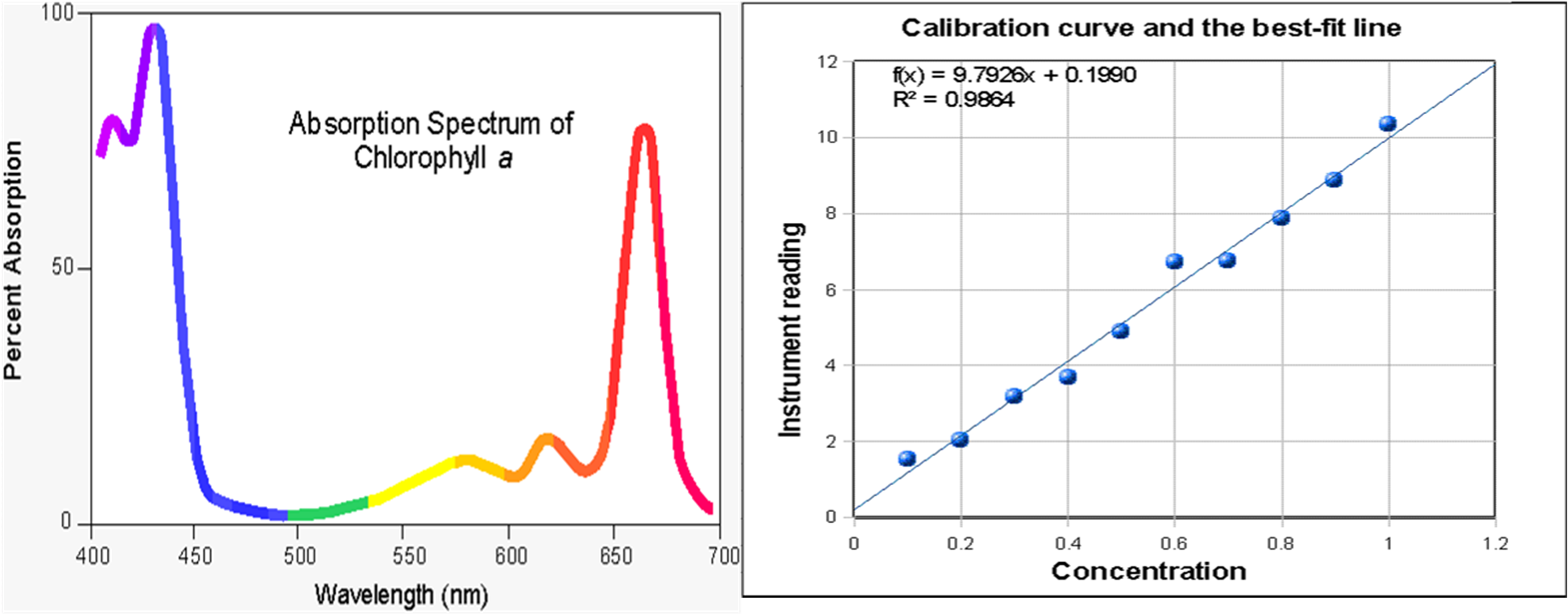
Typical absorbance (A) and reaction kinetic rate (B) for MB degradation... | Download Scientific Diagram
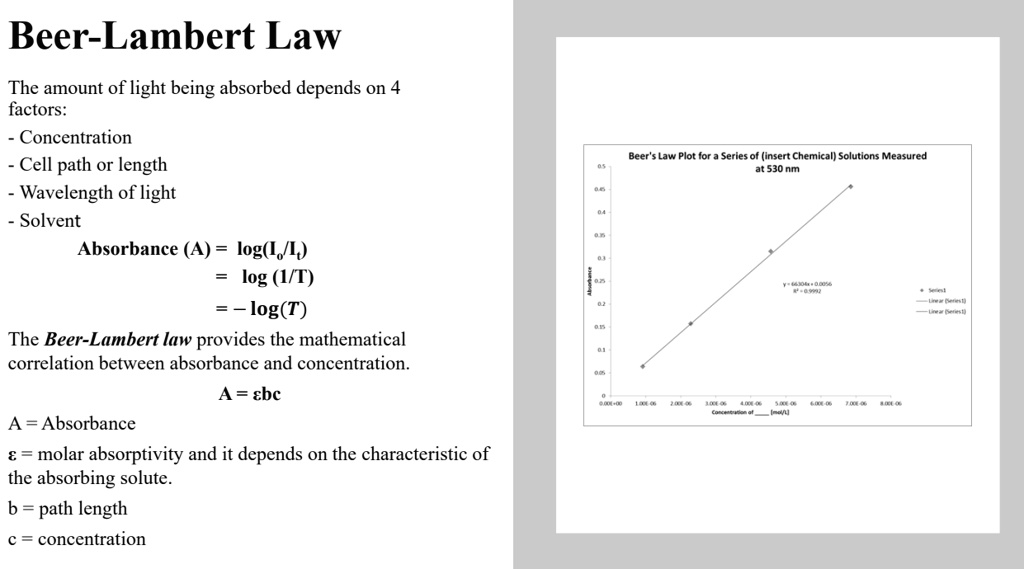
SOLVED: Beer-Lambert Law The amount of light being absorbed depends on 4 factors: Concentration Cell path or length Wavelength of light Solvent Absorbance (A) = log(LJI) log (IT) log(T) The Beer-Lambert law